RIBS 2025 Research Project
Genetic and Environmental Modulation of Vibrio fischeri Motility and Biofilm Formation
What This Was About
At RIBS 2025, I spent weeks working with Vibrio fischeri — the glowing bacteria that live inside Hawaiian bobtail squid.
- These bacteria form biofilms (sticky layers of cells) and move around using motility (like swimming).
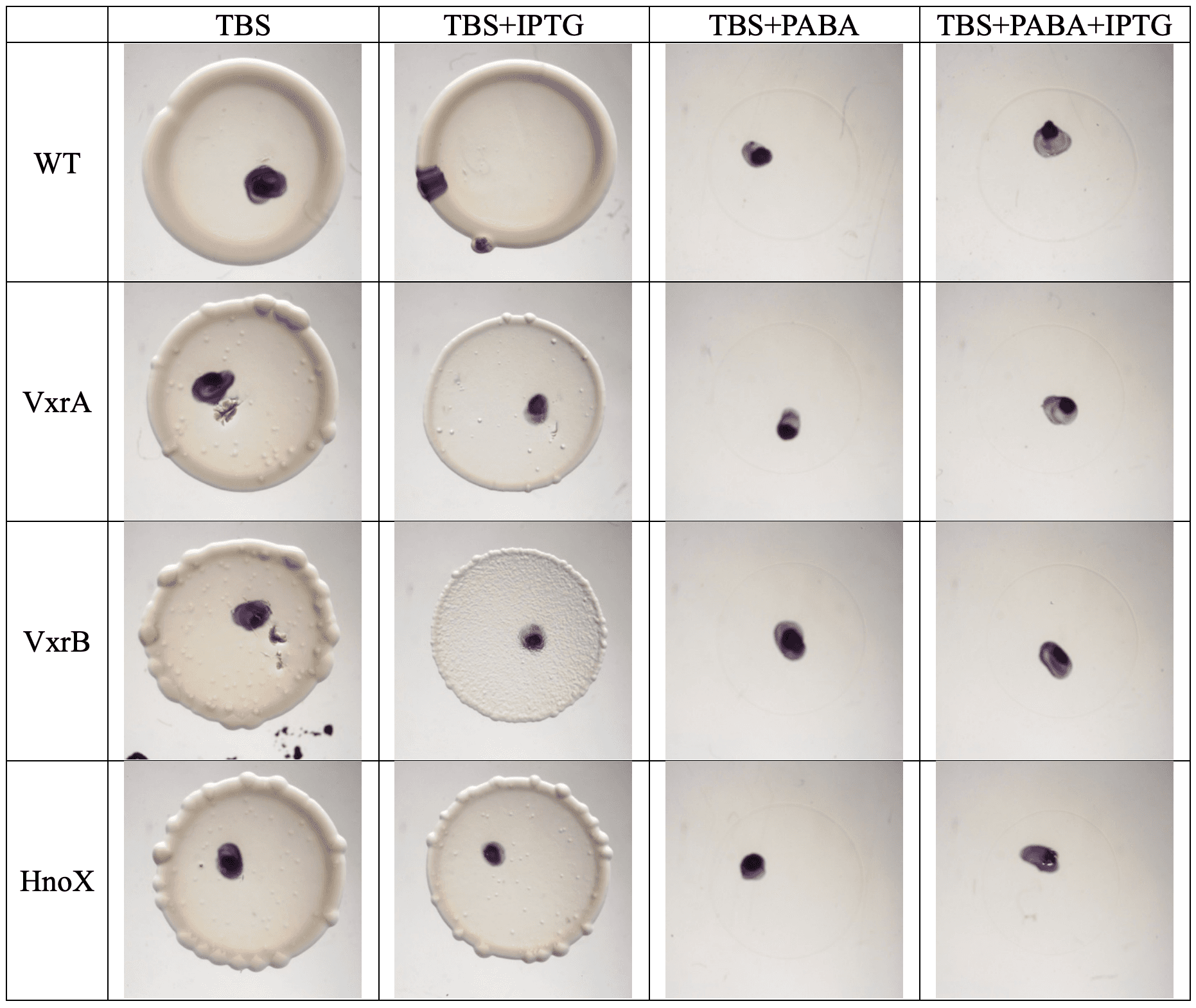
- We wanted to see how genes (like VxrA, VxrB, CrvY, BinK…) and environmental conditions (like calcium, PABA, antibiotics, temperature) affect those behaviors.
- To do this, we used mutant strains, CRISPRi knockdowns, and lots of phenotypic assays.
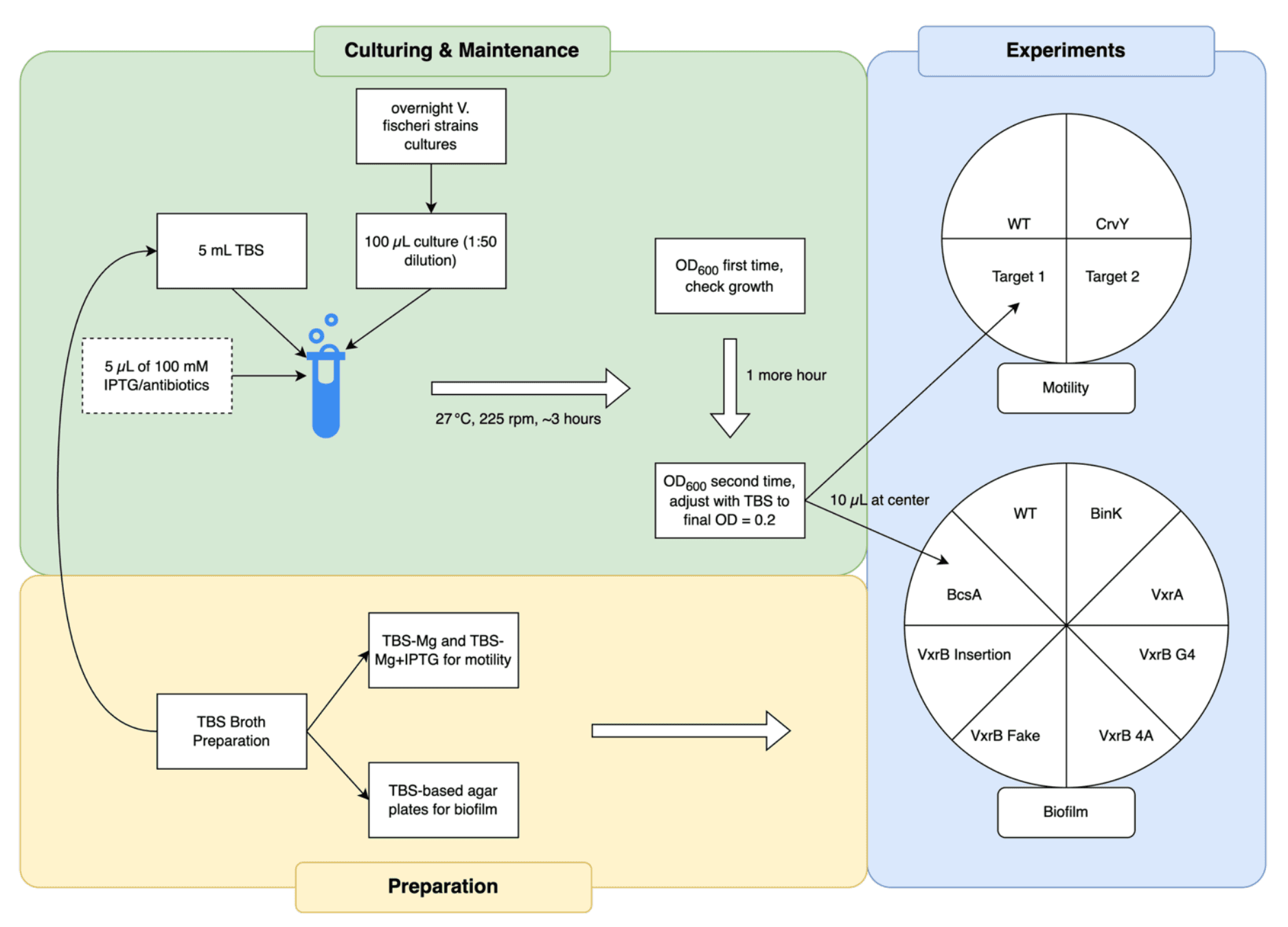
What I Actually Did
I was responsible for:
- Preparing different growth plates (TBS, TBS+CaCl₂, TBS+PABA, etc.)
- Running motility assays (measuring how far colonies spread in soft agar)
- Running biofilm assays (looking at smooth vs rough vs wrinkled colonies)
- Collecting and organizing all the data (lots of scanning plates and measuring colony diameters).
- Comparing our results to published studies (Dial et al. 2021, Septer & Visick 2024).
What We Found
Here’s the big picture from my experiments:
Motility
- Most strains looked normal.
- CrvY mutants swam a little less → ✔ matched what we expected.
- Antibiotics + IPTG? Only tiny changes, nothing dramatic.
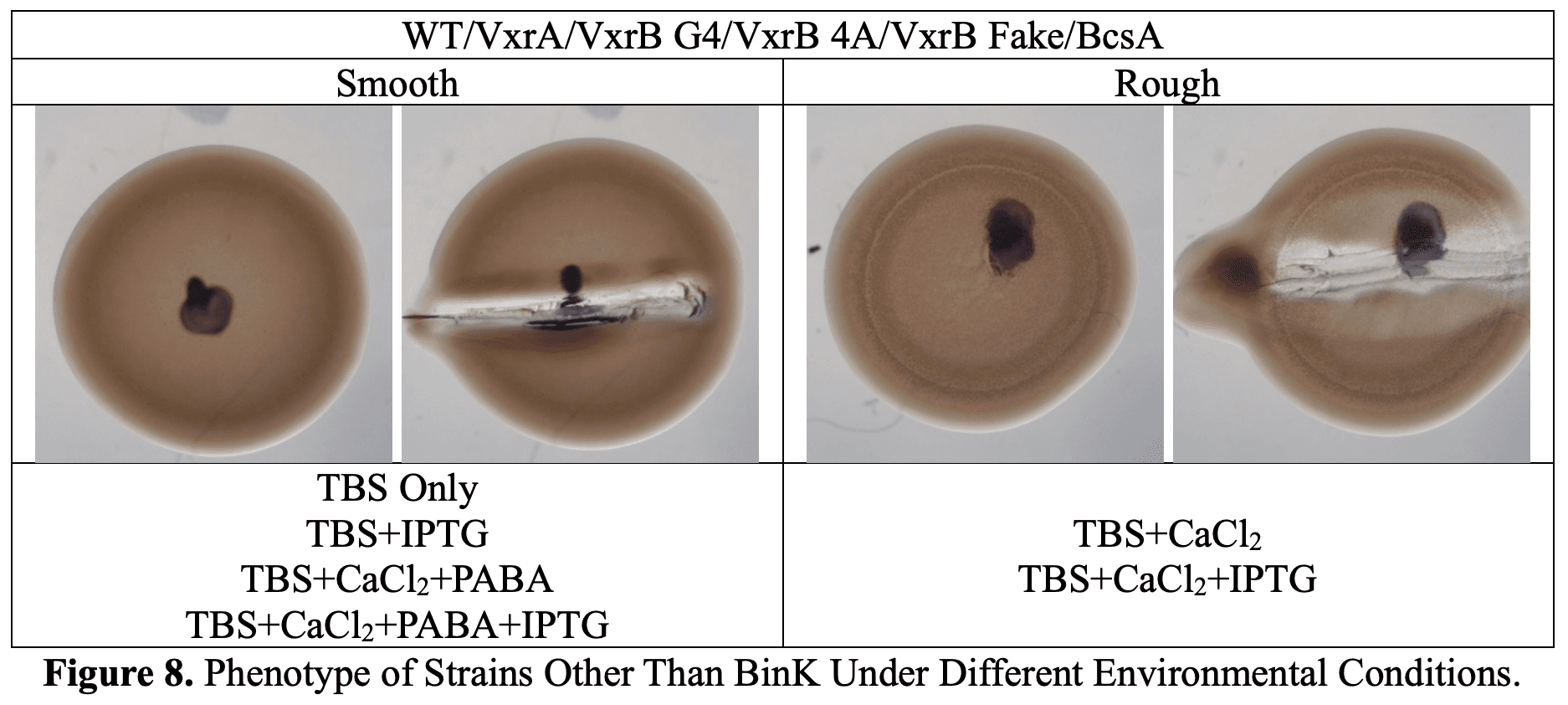
Biofilm Formation
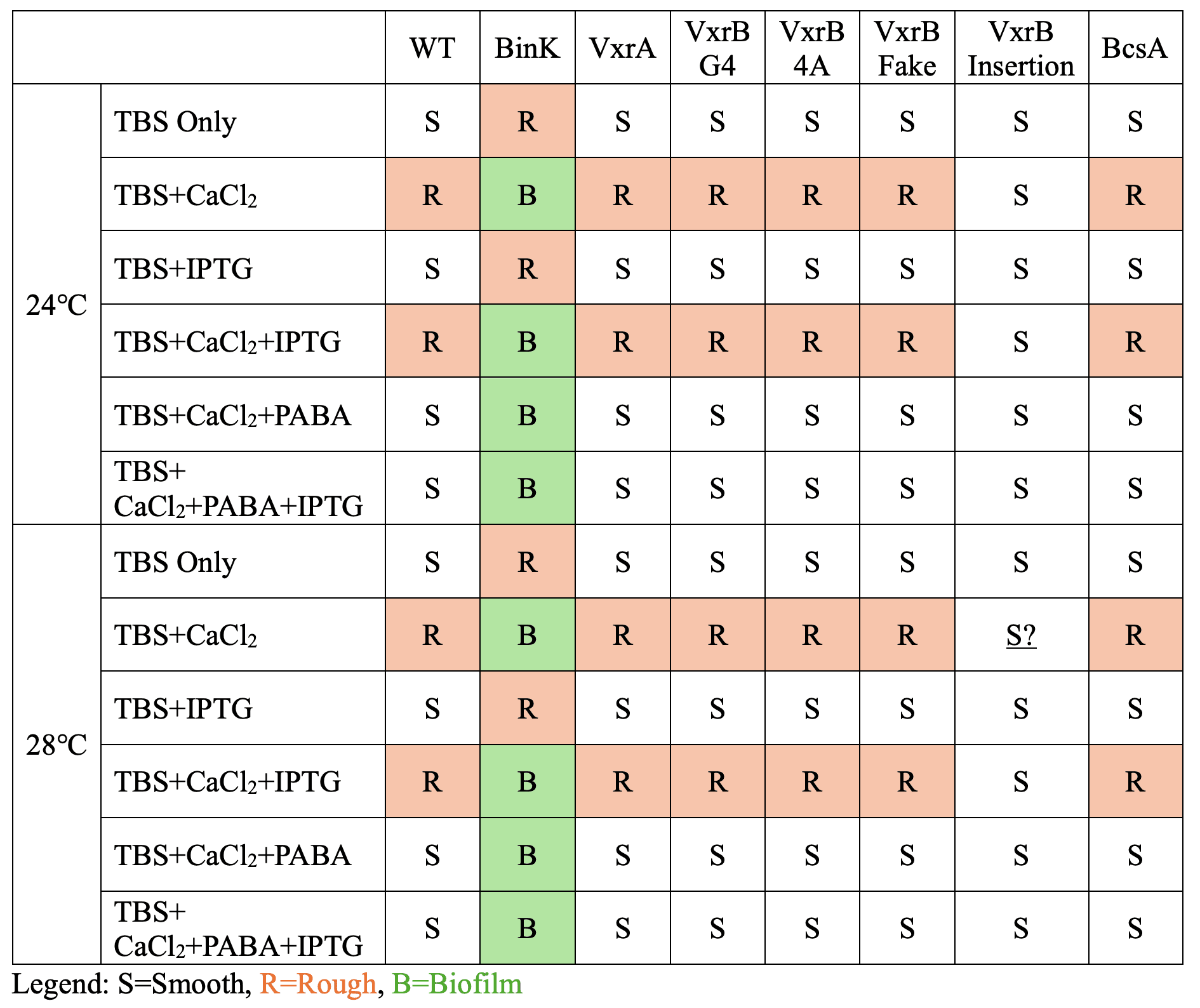
| Condition | BinK mutant | Other strains (WT, VxrA, VxrB variants, BcsA) |
|---|---|---|
| TBS only | ❌ smooth colonies | ❌ smooth colonies |
| + CaCl₂ | ✅ wrinkled biofilm | 😐 only rough, not full biofilm |
| + CaCl₂ + PABA | ❌ inhibited | ❌ inhibited |
| + IPTG | ❌ no big effect | ❌ no big effect |
Fun fact: The VxrB insertion mutant was the odd one out — it stayed smooth no matter what we tried.
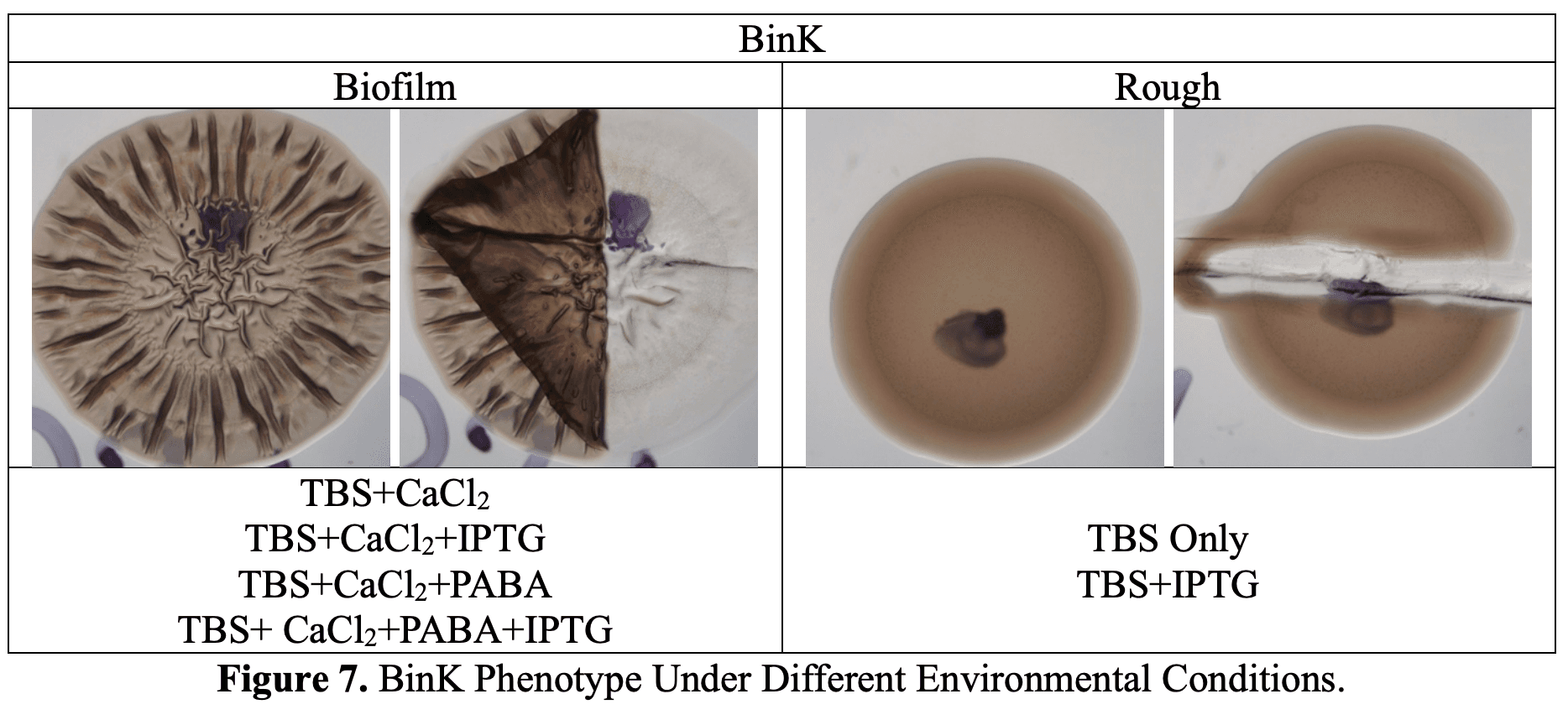
What I Learned
- Biofilms take patience. At first, our colonies didn’t look like “real” biofilms, but then I realized we only incubated for 48 hours. Published studies waited 72–96 hours.
- Environmental signals matter a lot:
- Calcium = boosts biofilm
- PABA = suppresses biofilm
- Our triparental mating experiments failed — which taught me that sometimes in science, not getting results is still a result.
Takeaways
This project taught me:
- Real research isn’t always neat — experiments fail, timelines are short, and you have to troubleshoot constantly.
- I got hands-on with microbiology techniques (plate pouring, sterile culturing, CRISPRi strains).
- I saw how genetics + environment = behavior in microbes.
- And most importantly: I discovered that I love digging into the “why” behind weird results (like our wrinkly VxrB strains).
Overall, even though we didn’t get perfect results, I’m proud of how much I contributed. From long hours plating and measuring colonies to troubleshooting failed conjugations, I really got a taste of what microbiology research is like — and I’d do it all again in a heartbeat.
